'He nearly left us twice': Mother’s plea for RSV vaccine after baby’s fight for life
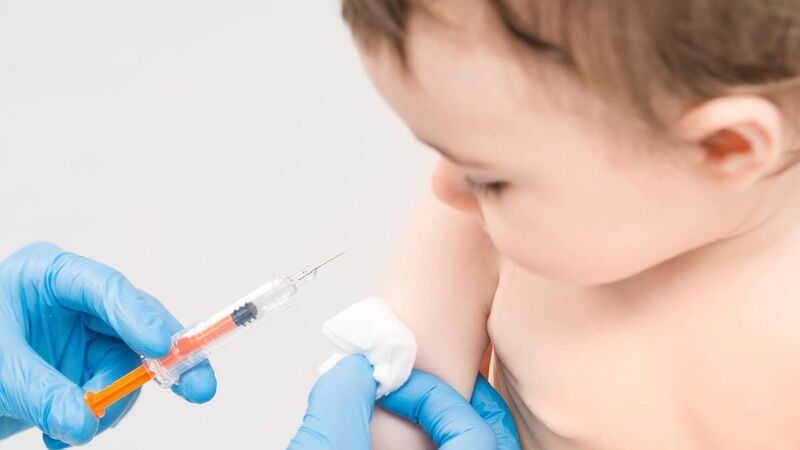
The HSE and Department of Health have confirmed the RSV pilot continues this coming winter, while a final decision is awaited beyond that.
In 2023, Lynn Power’s baby Rian was on a ventilator for 10 days because of RSV as doctors battled to save him.
Today, he still faces related health struggles.
Among the difficult memories she and her husband have is following an ambulance, lights flashing, from the hospital in Waterford to Dublin for specialist care.
It all came back to her this week when Hiqa said a pilot RSV immunisation programme running since 2024 was safe and effective but too expensive at current prices.
“He’s two and a half now and he’s come so far. He’s on the up now but it has been a long, long road to get him to where he is now,” Ms Power said.
“He’s doing very well now but we’re so lucky to have him. He was ventilated, he nearly left us twice.”
Rian was born in early November and became unwell about two weeks later. She went to their GP twice and the out-of-hours service but was not reassured.
When they went to the emergency department at University Hospital Waterford that Saturday evening, she said: “They admitted him straightaway.”
As his condition worsened, she said he was gasping for breath and was put on oxygen. She described watching him being put on a ventilator as “horrific”, though she knew it was life-saving. “There’s no other word for watching a child being intubated. I can’t say enough good things about the care that he got in Waterford.”
Later that day, a transfer team arrived from CHI at Temple Street, home to one of Ireland's two paediatric intensive care units.
Ms Power and her husband followed them to Dublin, leaving their oldest child being looked after at home.
She remembers sitting by his incubator for hours. “There was a certain machine and you’re watching it, you hear the beep and you know he’s ok.” He remained on the ventilator for days until "he turned the corner and we don’t know why".
Rian was transferred back to University Hospital Waterford about three weeks later, and was home for Christmas.
However, his mother said he had a lot of feeding issues and she was devastated he had come-down issues from the morphine after being ventilated and medicated for so long.
He remains under University Hospital Waterford for RSV-related complications, including lung injury.
“People think I over-react [about RSV risks] but until you walk that road, you don’t know,” she said.
She called on the HSE and health minister Jennifer Carroll MacNeill to support the immunisations.
“We are spending so much money on bringing teams to Waterford and back to Dublin, why not give this vaccine to babies to stop all this,” she said.
Hiqa was asked by the minister to evaluate the pilot for long term use.
Babies are given a monoclonal antibody called nirsevimab by injection if their parents consent. This is manufactured by Sanofi/AstraZeneca.
An estimated 1,800 children were admitted to hospital every winter with RSV before this.
Hiqa said the pilot programme “appears to have had a positive impact” after staff had been dealing with increased pressure previously.
It considered options including immunising babies or vaccinating mothers, as some countries do.
Both options are “safe”, it advised on Tuesday.
However, it noted costs were not transparent and potentially extremely high, adding “even when we take into account the money saved because fewer people have to go to their GP or are admitted to hospital or paediatric intensive care units".
Costs needed to be almost halved, it suggested, noting a range of €15.6m to €58.5m over five years, depending on the approach taken.
“Offering RSV immunisation to infants could be cost effective if the HSE can negotiate prices that are lower than those that are listed or reported internationally,” it concluded.
Doctors like Children’s Health Ireland clinical director and paediatric emergency medicine consultant Turlough Bolger see this differently however.
He warned: “It’s almost impossible to avoid RSV, and it’s very infectious”.
He acknowledged Hiqa weighed that against vaccine prices.
“That’s a commercial negotiation,” he said.
“So the most important thing is we look at the clinical effectiveness. And look at these children being transferred up from Cork, Galway or Donegal with severe bronchiolitis and RSV infection, and the trauma and the danger of having to drive all that distance up to an ICU bed.”
Finding the balance is challenging, according to health economist and senior lecturer at University College Cork Brian Turner.
“Essentially what Hiqa have said is ‘it would be great if we could do this but the price would need to be a lot lower in order for it to be cost effective’,” he said.
Hiqa analysed nine scenarios, he pointed out.
These assessments are “standard procedure” he said, adding: “It’s quite often the case that new drugs or devices are found to be not cost-effective at the quoted price, but then if the price is reduced they would be deemed cost-effective.
“So quite often then the HSE will enter into negotiations with the providers and come to an agreement.”
This comes down to what he described as “opportunity cost” in economic terms.
“Is this a good use of money or could it be put to better use in another area of the health services, that’s always the decision in the background,” he said. “If you spend it on this, you can’t spend it on a new cancer drug or heart stents or homecare.”
There were protests in 2022 after a breakdown in talks between the HSE and pharma company Vertex around children’s cystic fibrosis medication.
Shortly before this was resolved, doctors with the National Clinical Programme for Cystic Fibrosis wrote an open letter calling for partnership in care.
“This partnership is under threat, because of the approach of Vertex who we believe are effectively using this group of vulnerable children as pawns in negotiations for greater profit,” they said.
That evaluation was done by the National Centre for Pharmacoeconomics.
Professor Michael Barry, NCPE clinical director, told the evaluations in general are based on various factors.
Asked if protests have an impact, he said: “Obviously, we’re aware of this but it doesn’t feed into our assessment.
“Our assessments would be purely on the science, purely on the clinical evidence, clinical trials, real-world evidence and what is that saying to us, how does it match it up with the cost, is there any relationship with the price being charged and the outcomes.”
He added: “We don’t make the decision. We make a recommendation and that’s for others to decide then. So ultimately it comes down to the HSE to decide if they’re going to reimburse the drug.”
The HSE and Department of Health have confirmed the RSV pilot continues this coming winter, while a final decision is awaited beyond that.












