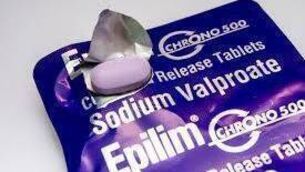
Father granted files on adverse effects of drug
Liam Grant, a Dublin-based accountant, is expected to receive details about adverse reactions to the drug filed in all 27 EU member states from the EMA.
“It’s great news as it will complement similar reports we already have from the US and UK,” said Mr Grant.
Lunchtime News
Newsletter
Keep up with stories of the day with our lunchtime news wrap and important breaking news alerts.