Dutch clinic stem cells ‘were not for human use’
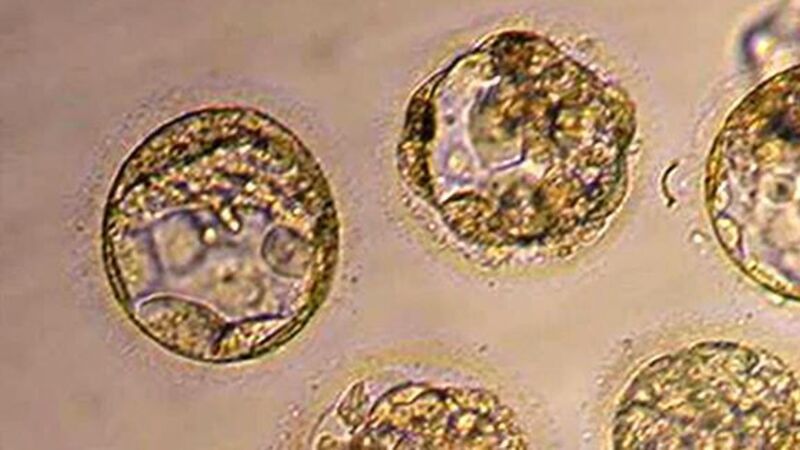
Dr Robert Trossel, aged 55, said he only discovered that a batch of vials sent to him in Rotterdam in 2006 carried the warning when he took part in a BBC Newsnight programme.
Dr Trossel also came to the attention of the Irish Medicines Board in 2006 when he began flying here to use Dr John Dunphy’s practice at Carrigaline, Co Cork, to provide such stem cell therapy.
Lunchtime News
Newsletter
Get a lunch briefing straight to your inbox at noon daily. Also be the first to know with our occasional Breaking News emails.












