Mother of boy with severe epilepsy says medicinal cannabis legislation is 'life-changing'
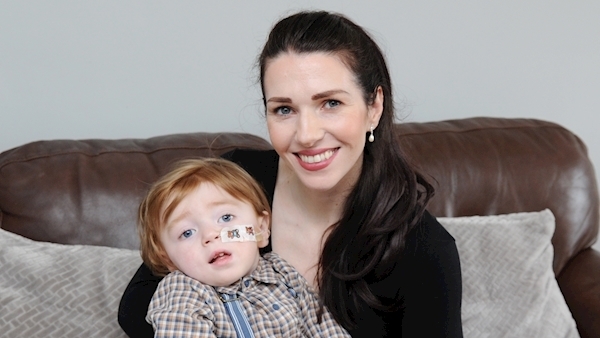
A mother who said she felt like a criminal after security at Dublin Airport seized her son's THC medicinal cannabis last December has described the new legislation for access to such products as "life-changing".
Health Minister Simon Harris today signed legislation which will allow for the operation of the Medical Cannabis Access Programme on a pilot basis for five years.
Noreen O'Neill, who lives in Wilton, Cork, said the move will give the gift of quality of life to her son, Michael, who turns three in August.
Michael suffered from intractable seizures linked to severe epilepsy for the first 16-months of his life. Standard medication didn't work for him.
He couldn't sit up, reach, roll, smile or lift his arms, legs or head. Noreen said he couldn't swallow properly and was at risk of drowning on his own saliva. He was fed through a tube in his nose.
He began taking CBD oil early last year and hasn't been in hospital in an emergency situation since. But as he grew, he needed the more potent THC medicinal cannabis which could only be sourced abroad.

Noreen was one of several mothers who pleaded for the law to change.
She described today as a "beautiful day" not just for her family, and those who have campaigned with her, but for all those who will be able to avail of medicinal cannabis here in the future.
"It's surreal really and I am still trying to process the news," she said.
Just when we thought we were in reach of it, it got pushed back, and it seemed for a long time that nothing was happening. But today is a great day.
"Now, we can try to start reclaiming what Michael lost in the first year of his life. He missed a lot of development. But now he's going to creche, he's horse-riding and swimming. It's like a different life.
"So many people deserve praise and thanks for what's happened. As long as you're fighting for the right thing, the tables will always turn your way."
Mr Harris described the signing of legislation as “significant milestone”, and said his Department took its time before launching the programme to make sure they got it right.
“I've met with a number of European counterparts over the last couple of years in relation to this and different countries are trying to grapple with this issue,” Mr Harris said.
“It's very important we get this right, it's very important we follow clinical advice, and it's very important that we have clinical buy-in. At the end of the day, these are clinical decisions. This is about the prescribing decisions that are made by doctors in our hospitals when they meet patients in relation to certain conditions. So we took our time to get this right, and I think we have,” he said.
Medical consultants will be able to prescribe approved cannabis-based products to patients in cases where they have failed to respond to standard treatments for:
The next step will see suppliers of medicinal cannabis products apply to the Health Products Regulatory Authority to have their medicines added to the approved list.
People Before Profit TD Gino Kenny, who has long campaigned for medicinal cannabis access for patients, welcomed the legislation and pilot programme.
“I would however say that the programme, as the government have laid out, is quite restrictive in that it does not include a number of conditions that could derive benefit from medicinal cannabis, most notably chronic pain,” he said.
Your questions about medicinal cannabis answered
Health Minister Simon Harris has signed legislation which will allow for the operation of a Medical Cannabis Access Programme on a pilot basis for five years.
The scheme will allow some patients access to cannabis-based products.
Anyone who suffers from a specific condition, and has a prescription from a medical consultant, will be able to apply for a listed cannabis-based treatment that has been approved under the scheme.
Those who experience spasticity associated with multiple sclerosis, suffer intractable nausea and vomiting associated with chemotherapy, or have severe, refractory, treatment-resistant, epilepsy can apply for an approved medical cannabis product.
They will need a prescription from a consultant, who can only prescribe a cannabis-based product if the patient has failed to respond to standard treatments.
None at the moment. The first stage of the programme is for potential suppliers to apply to have their medical cannabis products assessed for suitability for medical use.
No medical cannabis products are available in Ireland at present, however, the legislation signed today now means that commercial operators whose cannabis products meet the specified requirements set out in the legislation and are listed will be able to supply to the Irish market.
The suppliers will apply to the Health Products Regulatory Authority to have a product considered for inclusion in the Schedule of ‘specified controlled drugs’ in this new piece of legislation.
Pharmacists will be able to dispense Cannabis for Medical Use to patients as set out in the legislation, on foot of a valid prescription.
However all this will take time. As we have noted, no medical cannabis products are available in Ireland at the moment and the programme is still in the first phase where suppliers will submit their products for consideration for use under the scheme.
Once suppliers make the specified controlled drugs available on the Irish market, they will be available under prescription from pharmacists.