A breast biopsy result in an instant
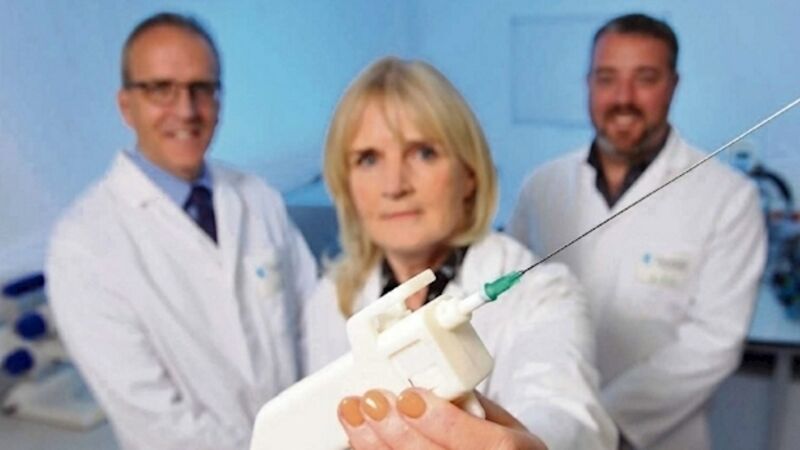
The SmartProbe needle can give women a cancer all-clear immediately, thus sparing them an anxious wait and reducing a test backlog clogged with benign results, says
A biopsy needle originally devised for safer anaesthesia may yet reduce the awful ordeal of waiting days, or even weeks, to find out if you have breast cancer.
Lunchtime News
Newsletter
Get a lunch briefing straight to your inbox at noon daily. Also be the first to know with our occasional Breaking News emails.