Explainer: What is the AstraZeneca vaccine supply row about?
A row has erupted between the European Union, the United Kingdom and pharmaceutical company AstraZeneca over the supply of coronavirus vaccines.
The EU is frustrated at supply shortages and has demanded AstraZeneca doses be sent from British plants to make up for a shortfall.
Here’s what we know about the dispute and its potential implications:
AstraZeneca has said initial deliveries to the EU will fall short because of a production glitch – said to be at a hub in Belgium – and it will not be able to meet its supply targets for the first three months of this year.
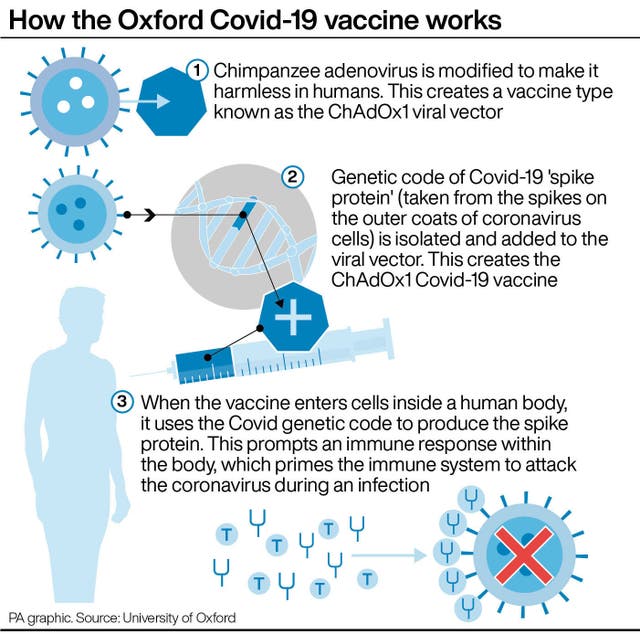
The Anglo-Swedish company announced initial deliveries in the EU would total approximately 31 million doses, rather than the anticipated 80 million in the first quarter of the year.
With the speed of the UK’s vaccine rollout outstripping other European countries, the EU has suggested doses produced in Europe have been directed elsewhere.
EU Commission president Ursula von der Leyen has now called for an explanation from AstraZeneca for delivery hold-ups, as she insisted the supply orders are “binding” and “the contract is crystal clear”.
EU health commissioner Stella Kyriakides has accused AstraZeneca of a “lack of clarity” and “insufficient explanations”, adding “the answers of the company have not been satisfactory” following a meeting on Monday.
She has proposed forcing all drug-makers to register their Covid-19 vaccine exports in advance, so the bloc can keep track of what they are doing.
Following talks on Wednesday, Ms Kyriakides added: “We regret the continued lack of clarity on the delivery schedule.”
Latvian foreign affairs minister Edgars Rinkevics said states could take AstraZeneca to court for breach of supply contracts if it does not honour its delivery schedule.
And Germany’s health minister Jens Spahn supported restrictions on vaccine exports, saying Europe should have its “fair share”.
He added: “I can understand that there are production problems but then it must affect everyone in the same way.”
The company’s chief executive Pascal Soriot said the contract only committed to meet the EU’s demands to its “best effort”.
In an interview with Italy’s la Repubblica newspaper that was published on Tuesday, he said the EU’s deliveries were delayed in part because the bloc signed its contract three months later than the UK, and therefore EU manufacturing facilities were still catching up
Translated by Politico, Mr Soriot reportedly said the “contract is very clear: Our commitment is, I am quoting, ‘our best effort'”.
He explained that AstraZeneca and its partner Oxford University had signed a deal with the UK Government for 100 million doses three months before the EU deal for 400 million doses was agreed.
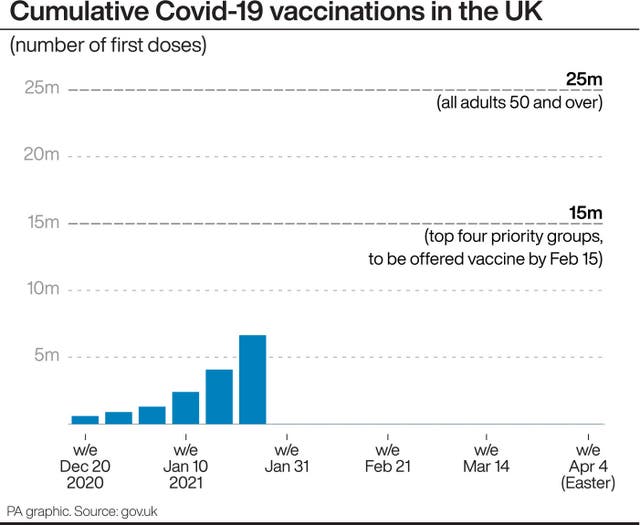
In response to the EU demanding their doses were shipped concurrently, Mr Soriot suggested it was a “super stretch goal”, and added: “We said, ‘Okay, we’re going to do our best, we’re going to try, but we cannot commit contractually because we are three months behind UK’.”
British Prime Minister Boris Johnson said this week he is “very confident” about the UK’s vaccine supply, while UK Cabinet Office minister Michael Gove insisted there “will be no interruption”.
Government vaccine tsar Nadhim Zahawi also said on Tuesday he is “confident” supply of the Pfizer jab – which is produced in Belgium – will continue.
Asked if the EU could prevent Pfizer vaccines from being exported, he told Sky News: “No, I’m confident that the Pfizer vaccine will be delivered.
“Pfizer have made sure that they have always delivered for us.
“They will continue to do so.”
The vaccine is expected to be approved today by the EMA.
The HSE's Chief Clinical Officer, Dr Colm Henry, said they are waiting for that EU advice before making decisions on use.
There are fears that Ireland's supply of the vaccine could be halved.
Dr Henry acknowledged today the concern expressed in Germany that relatively few older people had been included in the AstraZeneca trials which made it hard to draw confident conclusions, “but that doesn't mean that the vaccine isn't effective among older people and the EMA will consider the limited information available, but also how they can extrapolate from the results among younger people to older people - I would urge people, let's wait and see.
“We know what the absence of a vaccine means - we've seen that for the last 11 months and it doesn't look good.
"The vaccine is the one beacon of hope we have in 2021 for older and younger people.”





